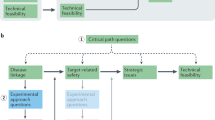
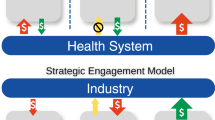
Abstract
A new model for translational research and drug repositioning has recently been established based on three-way partnerships between public funders, the pharmaceutical industry and academic investigators. Through two pioneering initiatives — one involving the Medical Research Council in the United Kingdom and one involving the National Center for Advancing Translational Sciences of the National Institutes of Health in the United States — new investigations of highly characterized investigational compounds have been funded and are leading to the exploration of known mechanisms in new disease areas. This model has been extended beyond these first two initiatives. Here, we discuss the progress to date and the unique requirements and challenges for this model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ashburn, T. T. & Thor, K. B. Drug repositioning: Identifying and developing new uses for existing drugs. Nat. Rev. Drug Discov. 3, 673–683 (2004).
Cook, D. et al. Lessons learned from the fate of AstraZeneca's drug pipeline: a five-dimensional framework. Nat. Rev. Drug Discov. 13, 419–431 (2014).
Mullard, A. Could pharma open its drug freezers? Nat. Rev. Drug Discov. 10, 399–400 (2011).
Carroll, J. Washington U gets second look at Pfizer molecules. Fierce Biotech [online], (2010).
Collins, F. S. Mining for therapeutic gold. Nat. Rev. Drug Discov. 10, 397 (2011).
LaMattina, J. The NIH is going to discover drugs... Really? Forbes [online], (2012).
Colvis, C. M. & Austin, C. P. The NIH-industry New Therapeutic Uses pilot program: demonstrating the power of crowdsourcing. Drug Repurp. Rescue Repos. 1, 15–16 (2015).
Southan, C., Williams, A. J. & Ekins, S. Challenges and recommendations for obtaining chemical structures of industry-provided repurposing candidates. Drug Discov. Today 18, 58–70 (2013).
Kaufman, A. C. et al. Fyn inhibition rescues established memory and synapse loss in Alzheimer mice. Ann. Neurol. 77, 953–971 (2015).
Strittmatter, S. M. Safety and tolerability of AZD0530 (Saracatinib) in Alzheimer's disease. ClinicalTrials.gov [online], (2014).
Acknowledgements
The UK Medical Research Council (MRC) and the US National Institute of Health (NIH) initiatives were supported by a large range of individuals within the MRC and the NIH as well as external investigators, and investigators from AstraZeneca and Medimmune. At the risk of excluding many, in particular the authors would like to thank J. Latimer (MRC), C. Colvis and B. Dunn (NIH–NCATS), G. Wilkinson, C. Wilks, A. Longton, S. Curran, K. Hickling and the members of the New Opportunities Emerging Innovations Unit (AstraZeneca). The authors would like to acknowledge the contribution made by all those involved and the exciting ideas and proposals received from academic investigators.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M.N.P., C.D.W., H.J.S., M.B. and K.J.E are employees of AstraZeneca, D.E.F. is an employee of Allergan and C.W. is an employee of the UK Medical Research Council.
Related links
FURTHER INFORMATION
PowerPoint slides
Supplementary information
Supplementary information S1 (table)
List of AstraZeneca compounds offered in the MRC, NIH and NRPB initiatives (PDF 84 kb)
Supplementary information S2 (table)
Example of an AstraZeneca compound summary used in the MRC 'mechanisms of disease' call for proposals (PDF 102 kb)
Rights and permissions
About this article
Cite this article
Frail, D., Brady, M., Escott, K. et al. Pioneering government-sponsored drug repositioning collaborations: progress and learning. Nat Rev Drug Discov 14, 833–841 (2015). https://doi.org/10.1038/nrd4707
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4707
This article is cited by
-
Drug repurposing: a systematic review on root causes, barriers and facilitators
BMC Health Services Research (2022)
-
Protocol paper: a multi-center, double-blinded, randomized, 6-month, placebo-controlled study followed by 12-month open label extension to evaluate the safety and efficacy of Saracatinib in Fibrodysplasia Ossificans Progressiva (STOPFOP)
BMC Musculoskeletal Disorders (2022)
-
Exploring new uses for existing drugs: innovative mechanisms to fund independent clinical research
Trials (2021)
-
Diarylthiazole and diarylimidazole selective COX-1 inhibitor analysis through pharmacophore modeling, virtual screening, and DFT-based approaches
Structural Chemistry (2019)
-
Importance of the novel organic cation transporter 1 for tyrosine kinase inhibition by saracatinib in rheumatoid arthritis synovial fibroblasts
Scientific Reports (2017)