Abstract
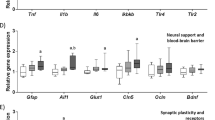
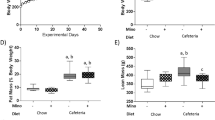
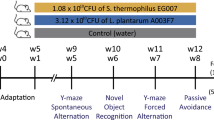
The western diet is known to have detrimental effects on cognition and the gut microbiota, but few studies have investigated how these may be related. Here, we examined whether a probiotic could prevent diet-induced memory deficits. Rats were pre-exposed to vehicle, low or high doses of VSL#3 for 2 weeks before half were switched from chow to a cafeteria diet (Caf) for 25 days; VSL#3 treatment continued until death. High-dose VSL#3 prevented the diet-induced memory deficits on the hippocampal-dependent place task, but the probiotic caused deficits on the perirhinal-dependent object task, irrespective of diet or dose. No differences were observed in anxiety-like behaviour on the elevated plus maze. Gut microbial diversity was dramatically decreased by Caf diet and here, VSL#3 was able to increase the abundance of some taxa contained in the probiotic such as Streptococcus and Lactobacillus and also other taxa including Butyrivibrio, which were decreased by the Caf diet. This affected the predicted profile of microbial metabolic pathways related to antioxidant and bile biosynthesis, and fat and carbohydrate metabolism. In the hippocampus, the Caf diet increased the expression of many genes related to neuroplasticity and serotonin receptor (5HT) 1A, which was normalised in Caf-High rats. Distance-based linear modelling showed that these genes were the best predictors of place memory, and related to microbiota principal component (PC) 1. Neuroplasticity genes in the perirhinal cortex were also affected and related to PC1 but object memory performance was correlated with perirhinal 5HT2C expression and microbiota PC3. These results show that probiotics can be beneficial in situations of gut dysbiosis where memory deficits are evident but may be detrimental in healthy subjects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Beilharz JE, Maniam J, Morris MJ . Short exposure to a diet rich in both fat and sugar or sugar alone impairs place, but not object recognition memory in rats. Brain Behav Immun 2014; 37: 134–141.
Kanoski SE, Davidson TL . Different patterns of memory impairments accompany short- and longer-term maintenance on a high-energy diet. J Exp Psychol Anim Behav Process 2010; 36: 313–319.
Jurdak N, Kanarek RB . Sucrose-induced obesity impairs novel object recognition learning in young rats. Physiol Behav 2009; 96: 1–5.
Beilharz JE, Maniam J, Morris MJ . Short-term exposure to a diet high in fat and sugar, or liquid sugar, selectively impairs hippocampal-dependent memory, with differential impacts on inflammation. Behav Brain Res 2016; 306: 1–7.
Hsu TM, Konanur VR, Taing L, Usui R, Kayser BD, Goran MI et al. Effects of sucrose and high fructose corn syrup consumption on spatial memory function and hippocampal neuroinflammation in adolescent rats. Hippocampus 2015; 25: 227–239.
Molteni R, Barnard RJ, Ying Z, Roberts CK, Gomez-Pinilla F . A high-fat, refined sugar diet reduces hippocampal brain-derived neurotrophic factor, neuronal plasticity, and learning. Neuroscience 2002; 112: 803–814.
Reichelt AC, Maniam J, Westbrook RF, Morris MJ . Dietary-induced obesity disrupts trace fear conditioning and decreases hippocampal reelin expression. Brain Behav Immun 2015; 43: 68–75.
Simpson HL, Campbell BJ . Review article: dietary fibre-microbiota interactions. Aliment Pharmacol Ther 2015; 42: 158–179.
Mayer EA, Knight R, Mazmanian SK, Cryan JF . Gut microbes and the brain: paradigm shift in neuroscience. J Neurosci 2014; 34: 15490–15496.
Burokas A, Moloney RD, Dinan TG, Cryan JF . Microbiota regulation of the Mammalian gut-brain axis. Adv Appl Microbiol 2015; 91: 1–62.
Frohlich EE, Farzi A, Mayerhofer R, Reichmann F, Jacan A, Wagner B et al. Cognitive impairment by antibiotic-induced gut dysbiosis: Analysis of gut microbiota-brain communication. Brain Behav Immun 2016; 56: 140–155.
Desbonnet L, Clarke G, Traplin A, O'Sullivan O, Crispie F, Moloney RD et al. Gut microbiota depletion from early adolescence in mice: Implications for brain and behaviour. Brain Behav Immun 2015; 48: 165–173.
Diaz Heijtz R, Wang S, Anuar F, Qian Y, Bjorkholm B, Samuelsson A et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci USA 2011; 108: 3047–3052.
Albenberg LG, Wu GD . Diet and the intestinal microbiome: associations, functions, and implications for health and disease. Gastroenterology 2014; 146: 1564–1572.
De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA 2010; 107: 14691–14696.
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M et al. Human gut microbiome viewed across age and geography. Nature 2012; 486: 222–227.
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014; 505: 559–563.
Devkota S, Wang Y, Musch MW, Leone V, Fehlner-Peach H, Nadimpalli A et al. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 2012; 487: 104–108.
Martinez-Medina M, Denizot J, Dreux N, Robin F, Billard E, Bonnet R et al. Western diet induces dysbiosis with increased E coli in CEABAC10 mice, alters host barrier function favouring AIEC colonisation. Gut 2014; 63: 116–124.
Turnbaugh PJ, Ridaura VK, Faith JJ, Rey FE, Knight R, Gordon JI . The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med 2009; 1: 6ra14.
Beilharz JE, Kaakoush NO, Maniam J, Morris MJ . The effect of short-term exposure to energy-matched diets enriched in fat or sugar on memory, gut microbiota and markers of brain inflammation and plasticity. Brain Behav Immun 2016; 57: 304–313.
Bruce-Keller AJ, Salbaum JM, Luo M, Blanchard ET, Taylor CM, Welsh DA et al. Obese-type gut microbiota induce neurobehavioral changes in the absence of obesity. Biol Psychiatry 2015; 77: 607–615.
McCusker RH, Kelley KW . Immune-neural connections: how the immune system's response to infectious agents influences behavior. J Exp Biol 2013; 216: 84–98.
Krabbe KS, Reichenberg A, Yirmiya R, Smed A, Pedersen BK, Bruunsgaard H . Low-dose endotoxemia and human neuropsychological functions. Brain Behav Immun 2005; 19: 453–460.
Takechi R, Pallebage-Gamarallage MM, Lam V, Giles C, Mamo JC . Nutraceutical agents with anti-inflammatory properties prevent dietary saturated-fat induced disturbances in blood-brain barrier function in wild-type mice. J Neuroinflamm 2013; 10: 73.
Bercik P, Denou E, Collins J, Jackson W, Lu J, Jury J et al. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology 2011; 141: 599–609, 609.e591-593.
Neufeld KM, Kang N, Bienenstock J, Foster JA . Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol Motil 2011; 23: 255–264, e119.
Brigidi P, Vitali B, Swennen E, Bazzocchi G, Matteuzzi D . Effects of probiotic administration upon the composition and enzymatic activity of human fecal microbiota in patients with irritable bowel syndrome or functional diarrhea. Res Microbiol 2001; 152: 735–741.
Venturi A, Gionchetti P, Rizzello F, Johansson R, Zucconi E, Brigidi P et al. Impact on the composition of the faecal flora by a new probiotic preparation: preliminary data on maintenance treatment of patients with ulcerative colitis. Aliment Pharmacol Ther 1999; 13: 1103–1108.
Madsen K, Cornish A, Soper P, McKaigney C, Jijon H, Yachimec C et al. Probiotic bacteria enhance murine and human intestinal epithelial barrier function. Gastroenterology 2001; 121: 580–591.
Kuhbacher T, Ott SJ, Helwig U, Mimura T, Rizzello F, Kleessen B et al. Bacterial and fungal microbiota in relation to probiotic therapy (VSL#3) in pouchitis. Gut 2006; 55: 833–841.
Ma X, Hua J, Li Z . Probiotics improve high fat diet-induced hepatic steatosis and insulin resistance by increasing hepatic NKT cells. J Hepatol 2008; 49: 821–830.
Li Z, Yang S, Lin H, Huang J, Watkins PA, Moser AB et al. Probiotics and antibodies to TNF inhibit inflammatory activity and improve nonalcoholic fatty liver disease. Hepatology 2003; 37: 343–350.
Distrutti E, O'Reilly JA, McDonald C, Cipriani S, Renga B, Lynch MA et al. Modulation of intestinal microbiota by the probiotic VSL#3 resets brain gene expression and ameliorates the age-related deficit in LTP. PLoS ONE 2014; 9: e106503.
D'Mello C, Ronaghan N, Zaheer R, Dicay M, Le T, MacNaughton WK et al. Probiotics improve inflammation-associated sickness behavior by altering communication between the peripheral immune system and the brain. J Neurosci 2015; 35: 10821–10830.
Beilharz JE, Kaakoush NO, Maniam J, Morris MJ . The effect of short-term exposure to energy-matched diets enriched in fat or sugar on memory, gut microbiota and markers of brain inflammation and plasticity. Brain Behav Immun 2016; 57: 304–313.
Jeong JJ, Kim KA, Ahn YT, Sim JH, Woo JY, Huh CS et al. Probiotic Mixture KF Attenuates Age-Dependent Memory Deficit and Lipidemia in Fischer 344 Rats. J Microbiol Biotechnol 2015; 25: 1532–1536.
Aggleton JP, Brown MW . Contrasting hippocampal and perirhinal cortex function using immediate early gene imaging. Q J Exp Psychol B 2005; 58: 218–233.
Wan H, Aggleton JP, Brown MW . Different contributions of the hippocampus and perirhinal cortex to recognition memory. J Neurosci 1999; 19: 1142–1148.
Boitard C, Cavaroc A, Sauvant J, Aubert A, Castanon N, Laye S et al. Impairment of hippocampal-dependent memory induced by juvenile high-fat diet intake is associated with enhanced hippocampal inflammation in rats. Brain Behav Immun 2014; 40: 9–17.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001; 25: 402–408.
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB et al. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 2009; 75: 7537–7541.
Clarke KR . Non-parametric multivariate analyses of changes in community structure. Austr J Ecol 1993; 18: 117–143.
Langille MG, Zaneveld J, Caporaso JG, McDonald D, Knights D, Reyes JA et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol 2013; 31: 814–821.
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS et al. Metagenomic biomarker discovery and explanation. Genome Biol 2011; 12: R60.
Magnusson KR, Hauck L, Jeffrey BM, Elias V, Humphrey A, Nath R et al. Relationships between diet-related changes in the gut microbiome and cognitive flexibility. Neuroscience 2015; 300: 128–140.
Pyndt Jorgensen B, Hansen JT, Krych L, Larsen C, Klein AB, Nielsen DS et al. A possible link between food and mood: dietary impact on gut microbiota and behavior in BALB/c mice. PLoS ONE 2014; 9: e103398.
Davari S, Talaei SA, Alaei H, Salami M . Probiotics treatment improves diabetes-induced impairment of synaptic activity and cognitive function: behavioral and electrophysiological proofs for microbiome-gut-brain axis. Neuroscience 2013; 240: 287–296.
Frohlich EE, Farzi A, Mayerhofer R, Reichmann F, Jacan A, Wagner B et al. Cognitive impairment by antibiotic-induced gut dysbiosis: Analysis of gut microbiota-brain communication. Brain Behav Immun 2016; 56: 140–155.
Benton D, Williams C, Brown A . Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur J Clin Nutr 2007; 61: 355–361.
Stranahan AM, Norman ED, Lee K, Cutler RG, Telljohann RS, Egan JM et al. Diet-induced insulin resistance impairs hippocampal synaptic plasticity and cognition in middle-aged rats. Hippocampus 2008; 18: 1085–1088.
Leffa DD, Valvassori SS, Varela RB, Lopes-Borges J, Daumann F, Longaretti LM et al. Effects of palatable cafeteria diet on cognitive and noncognitive behaviors and brain neurotrophins' levels in mice. Metab Brain Dis 2015; 30: 1073–1082.
Sharma S, Fernandes MF, Fulton S . Adaptations in brain reward circuitry underlie palatable food cravings and anxiety induced by high-fat diet withdrawal. Int J Obes (Lond) 2013; 37: 1183–1191.
Sharma S, Fulton S . Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. Int J Obes (Lond) 2013; 37: 382–389.
Popoff MR, Poulain B . Bacterial toxins and the nervous system: neurotoxins and multipotential toxins interacting with neuronal cells. Toxins 2010; 2: 683–737.
Block ML, Zecca L, Hong JS . Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 2007; 8: 57–69.
de La Serre CB, Ellis CL, Lee J, Hartman AL, Rutledge JC, Raybould HE . Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am J Physiol Gastrointest Liver Physiol 2010; 299: G440–G448.
Duryee MJ, Willis MS, Schaffert CS, Reidelberger RD, Dusad A, Anderson DR et al. Precision-cut liver slices from diet-induced obese rats exposed to ethanol are susceptible to oxidative stress and increased fatty acid synthesis. Am J Physiol Gastrointest Liver Physiol 2014; 306: G208–G217.
Galter D, Unsicker K . Sequential activation of the 5-HT1(A) serotonin receptor and TrkB induces the serotonergic neuronal phenotype. Mol Cell Neurosci 2000; 15: 446–455.
Roshchina VV . Evolutionary considerations of neurotransmitters in microbial, plant, and animal cells. In: Lyte M, Freestone PEP (eds). Microbial Endocrinology: Interkingdom Signaling in Infectious Disease and Health. Springer New York: New York, NY, USA, 2010 pp 17–52.
Ogren SO, Eriksson TM, Elvander-Tottie E, D'Addario C, Ekstrom JC, Svenningsson P et al. The role of 5-HT(1A) receptors in learning and memory. Behav Brain Res 2008; 195: 54–77.
Jenkins TA, Nguyen JCD, Polglaze KE, Bertrand PP . Influence of tryptophan and serotonin on mood and cognition with a possible role of the gut-brain axis. Nutrients 2016; 8: 1.
Vauzour D, Vafeiadou K, Rodriguez-Mateos A, Rendeiro C, Spencer JP . The neuroprotective potential of flavonoids: a multiplicity of effects. Genes Nutr 2008; 3: 115–126.
Moura FA, de Andrade KQ, dos Santos JC, Goulart MO . Lipoic acid: its antioxidant and anti-inflammatory role and clinical applications. Curr Top Med Chem 2015; 15: 458–483.
Carlier JP, Bedora-Faure M, K'Ouas G, Alauzet C, Mory F . Proposal to unify Clostridium orbiscindens Winter et al. 1991 and Eubacterium plautii (Seguin 1928) Hofstad and Aasjord 1982, with description of Flavonifractor plautii gen. nov., comb. nov., and reassignment of Bacteroides capillosus to Pseudoflavonifractor capillosus gen. nov., comb. nov. Int J Syst Evol Microbiol 2010; 60: 585–590.
Kaakoush NO, Martire SI, Raipuria M, Mitchell HM, Nielsen S, Westbrook RF et al. Alternating or continuous exposure to cafeteria diet leads to similar shifts in gut microbiota compared to chow diet. Mol Nutr Food Res 2016; 61: 1.
Acknowledgements
This work was supported in part by funding from NHMRC (Application number: 1023073) and UNSW Sydney to MJM. Some project support, including the probiotic, was donated by the distributor of the product. This company had no role in the study design, data analyses or interpretation and has no ownership of the data. NOK is supported by a Cancer Institute NSW Career Development Fellowship (15/CDF/1-11) and JEB is supported by an Australian Postgraduate Award scholarship. We would like to thank Gleiciane Soares for blind scoring the behavioural data.
Author contributions
Conceived and designed experiments: JEB, MJM. Performed the experiments: JEB. Behavioural data: JEB. Brain data: JEB, JM. Microbiota data: JEB, NOK. Wrote the paper and interpreted data: JEB, NOK, JM, MJM. All authors approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Rights and permissions
About this article
Cite this article
Beilharz, J., Kaakoush, N., Maniam, J. et al. Cafeteria diet and probiotic therapy: cross talk among memory, neuroplasticity, serotonin receptors and gut microbiota in the rat. Mol Psychiatry 23, 351–361 (2018). https://doi.org/10.1038/mp.2017.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2017.38
This article is cited by
-
Investigation of memory-enhancing effects of Streptococcus thermophilus EG007 in mice and elucidating molecular and metagenomic characteristics using nanopore sequencing
Scientific Reports (2022)
-
Fecal microbiota transplantation as a promising treatment option for osteoporosis
Journal of Bone and Mineral Metabolism (2022)
-
Modulating role of serotonergic signaling in sleep and memory
Pharmacological Reports (2022)
-
Intermittent cafeteria diet identifies fecal microbiome changes as a predictor of spatial recognition memory impairment in female rats
Translational Psychiatry (2020)
-
Mid-life microbiota crises: middle age is associated with pervasive neuroimmune alterations that are reversed by targeting the gut microbiome
Molecular Psychiatry (2020)