Abstract
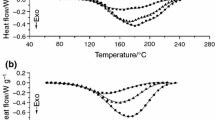
The thermal stability of latent resin systems, cycloaliphatic epoxy/4,4′-dihydroxydiphenylsulfone/aluminum complexes, was investigated by dynamic differential scanning calorimetry (DSC) analysis. Experiments were conducted under non-isothermal condition in a nitrogen atmosphere at the heating rate of 10, 20, 30 and 40 °C/min, respectively. TG curves showed that, in the temperature range of 25 to 600 °C, the stability of the resin systems could be enhanced by increasing the length of the aliphatic chain in the initiator. Both the Kissinger method and the Ozawa-Flynn-Wall method were employed to calculate activation energies of the decomposition reaction, and the values obtained from the two methods were compared. Moreover, the corresponding reaction mechanism was identified by the Achar differential method and the Coats-Redfern integral method. The experimental results showed that these four methods were reliable and effective to study the kinetics of the thermal decomposition reaction; and the most probable thermal decomposition mechanism of the resin systems we proposed was found to comply with Mampel power law (m=1).
Similar content being viewed by others
References
Baller J, Becker N, Ziehmer M, et al. Interactions between Silica Nanoparticles and an Epoxy Resin before and during Network Formation[J]. Polymer, 2009, 50(14): 3 211–3 219
Yang JP, Chen ZK, Fu SY, et al. Simultaneous Improvements in the Cryogenic Tensile Strength, Ductility and Impact Strength of Epoxy Resins by a Hyperbranched Polymer[J]. Polymer, 2008, 49(13–14): 3 168–3 175
Wang X, Hu Y, Song L. Flame Retardancy and Thermal Degradation Mechanism of Epoxy Resin Composites Based on a DOPO Substituted Organophosphorus Oligomer[J]. Polymer, 2010, 51(11): 2 435–2 445
Murai S, Fujieda S, Hayase S. A Novel Latent Initiator for Cationic Polymerization of Epoxide: Composite Catalysts Containing Aluminum Complexes Phase-Seperated in Epoxides[J]. Journal of Applied Polymer Science, 2002, 83(5):1 046–1 053
Murai S, Nakano Y, Hayase S. Novel Latent Initiator for Cationic Polymerization of Epoxides[J]. Journal of Applied Polymer Science, 2001, 80(2): 181–187
Barral L, Díez FJ, García-Garabal S. Thermodegradation Kinetics of a Hybrid Inorganic-Organic Epoxy System[J]. European Polymer Journal, 2005, 41(7):1 662–1 666
Ren H, Sun JZ, Zhao Q, et al. Synthesis and Characterization of a Novel Heat Resistant Epoxy Resin Based on N,N′-bis(5-hydroxy-1-naphthyl)Pyromellitic Diimide[J]. Polymer, 2008, 49(24): 5 249–5 253
Budrugeac P, Segal E. Application of Isoconversional and Multivariate Non-linear Regression Methods for Evaluation of the Degradation Mechanism and Kinetic Parameters of an Epoxy Resin[J]. Polymer Degradation and Stability, 2008, 93(6): 1 073–1 080
Liu WB, Qiu QH, Wang J. Curing Kinetics and Properties of Epoxy Resin-Fluorenyl Diamine Systems[J]. Polymer, 2008, 49(19): 4 399–4 405
Sun JT, Huang YD, Gong GF, et al. Thermal Degradation Kinetics of Poly(methylphenylsiloxane) Containing Methacryloyl Groups[J]. Polymer Degradation and Stability, 2006, 91(2): 339–346
Vincent L, Mija A, Sbirrazzuoli N. Liquid Crystalline and Isotropic Epoxy Thermosets: Mechanism and Kinetics of Non-isothermal Degradation[J]. Polymer Degradation and Stability, 2007, 92(11): 2 051–2 057
Brown ME, Maciejewski M, Vyazovkin S. Computational Aspects of Kinetics Analysis[J]. Thermochim Acta, 2000, 355: 125–143
Kissinger HE. Reaction Kinetics in Differential Thermal Analysis[J]. Analytical Chemistry, 1957, 29(11): 1 702–1 706
Salla JM, Ramis X. Comparative Study of the Cure Kinetics of an Unsaturated Polyester Resin Using Different Procedures[J]. Polymer Engineering and Science, 1996, 36(6): 835–851
Liang HB, Ding J, Shi WF. Kinetics and Mechanism of Thermal Oxidative Degradation of UV Cured Epoxy Acrylate/Phosphate Triacrylate Blends[J]. Polymer Degradation and Stability, 2004, 86(2): 217–223
Ozawa T. A New Method of Analyzing Thermogravimetric Data[J]. Bulletin of the Chemical Society of Japan, 1965, 38: 1 881–1 886
Flynn JH, Wall LA. Quick A. Direct Method for the Determination of Activation Energy from Thermogravimetric Data[J]. Journal of Polymer Science Part B: Polymer Letters, 1966, 4: 323–328
Chunxiu G, Yufang S, Donghua C. Comparative Method to Evaluate Reliable Kinetic Triplets of Thermal Decomposition Reactions[J]. Journal of Thermal Analysis and Calorimetry, 2004, 76: 203–216
Coats AW, Redfern JP. Kinetic Parameters from the Thermogravimetric Data[J]. Nature, 1964, 201: 68–69
Tomaszewicz E, Kotfica M. Mechanism and Kinetics of Thermal Decomposition of Nickel(II) Sulfate(VI) Hexahydrate[J]. Journal of Thermal Analysis and Calorimetry, 2004, 77(1): 25–31
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China (Nos.50973023 and 50773016), the Program for New Century Excellent Talents in University(No. NCET-09-0060), and the Aeronautical Science Foundation of China(No.2011ZF77010)
Rights and permissions
About this article
Cite this article
Wu, S., Liu, S., Liu, Y. et al. Thermal stability, thermal decomposition and mechanism analysis of cycloaliphatic epoxy/4,4′-dihydroxydiphenylsulfone/aluminum complexes latent resin systems. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 27, 1061–1067 (2012). https://doi.org/10.1007/s11595-012-0601-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-012-0601-5