Abstract
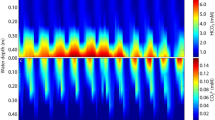
Two freshwater macrophytes, Ottelia alismoides and O. acuminata, were grown at low (mean 5 μmol L−1) and high (mean 400 μmol L−1) CO2 concentrations under natural conditions. The ratio of PEPC to RuBisCO activity was 1.8 in O. acuminata in both treatments. In O. alismoides, this ratio was 2.8 and 5.9 when grown at high and low CO2, respectively, as a result of a twofold increase in PEPC activity. The activity of PPDK was similar to, and changed with, PEPC (1.9-fold change). The activity of the decarboxylating NADP-malic enzyme (ME) was very low in both species, while NAD-ME activity was high and increased with PEPC activity in O. alismoides. These results suggest that O. alismoides might perform a type of C4 metabolism with NAD-ME decarboxylation, despite lacking Kranz anatomy. The C4-activity was still present at high CO2 suggesting that it could be constitutive. O. alismoides at low CO2 showed diel acidity variation of up to 34 μequiv g−1 FW indicating that it may also operate a form of crassulacean acid metabolism (CAM). pH-drift experiments showed that both species were able to use bicarbonate. In O. acuminata, the kinetics of carbon uptake were altered by CO2 growth conditions, unlike in O. alismoides. Thus, the two species appear to regulate their carbon concentrating mechanisms differently in response to changing CO2. O. alismoides is potentially using three different concentrating mechanisms. The Hydrocharitaceae have many species with evidence for C4, CAM or some other metabolism involving organic acids, and are worthy of further study.




Similar content being viewed by others
Abbreviations
- AMP:
-
Adenosine monophosphate
- ATP:
-
Adenosine triphosphate
- Alk:
-
Alkalinity
- CAM:
-
Crassulacean acid metabolism
- CCM:
-
Carbon dioxide concentrating mechanism
- DIC:
-
Dissolved inorganic carbon
- DTT:
-
Dithiothreitol
- FW:
-
Fresh weight
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- LDH:
-
Lactate dehydrogenase
- MDH:
-
Malate dehydrogenase
- NAD(P)-ME:
-
NAD(P)-malic enzyme
- OAA:
-
Oxaloacetate
- PEP:
-
Phosphoenol pyruvate
- PEPC:
-
PEP carboxylase
- PEPCK:
-
PEP carboxykinase
- PGK:
-
Phosphoglycerate kinase
- PPDK:
-
Pyruvate phosphate dikinase
- RuBisCO:
-
Ribulose 1,5-bisphosphate carboxylase–oxygenase
- RuBP:
-
Ribulose 1,5-bisphosphate
References
Aubry S, Brown NJ, Hibberd JM (2011) The role of proteins in C3 plants prior to their recruitment into the C4 pathway. J Exp Bot 62:3049–3059
Beer S (1989) Photosynthesis and photorespiration of marine angiosperms. Aquat Bot 34:153–166
Black MA, Maberly SC, Spence DHN (1981) Resistances to carbon dioxide fixation in 4 submerged freshwater macrophytes. New Phytol 89:557–568
Bowes G (2011) Single-cell C4 photosynthesis in aquatic plants. In: Raghavendra AS, Sage RF (eds) C4 photosynthesis and related CO2 concentrating mechanisms, vol 32. Advances in photosynthesis and respiration. Springer, Dordrecht, pp 63–80
Bowes G, Ogren WL, Hageman RH (1971) Phosphoglycolate production catalyzed by ribulose diphosphate carboxylase. Biochem Biophys Res Commun 45:716–722
Bowes G, Rao SK, Estavillo GM, Reiskind JB (2002) C4 mechanisms in aquatic angiosperms: comparisons with terrestrial C4 systems. Funct Plant Biol 29:379–392
Bradford MM (1976) Rapid and sensitive method for quantitation of microgram quantities of protein utilizing principle of protein-dye binding. Anal Biochem 72:248–254
Brain RA, Solomon KR (2007) A protocol for conducting 7-day daily renewal tests with Lemna gibba. Nat Protoc 2:979–987
Brown JMA, Dromgoole FI, Towsey MW, Browse J (1974) Photosynthesis and photorespiration in aquatic macrophytes. R Soc N Z Bull 12:243–249
Browse JA, Dromgoole FI, Brown JMA (1977) Photosynthesis in aquatic macrophyte Egeria densa. 1. CO2-14 fixation at natural CO2 concentrations. Aust J Plant Physiol 4:169–176
Casati P, Lara MV, Andreo CS (2000) Induction of a C4-like mechanism of CO2 fixation in Egeria densa, a submersed aquatic species. Plant Physiol 123:1611–1621
Chen L-Y, Chen J-M, Gituru RW, Wang Q-F (2012) Generic phylogeny, historical biogeography and character evolution of the cosmopolitan aquatic plant family Hydrocharitaceae. BMC Evol Biol 12:30
Cole JJ, Prairie YT, Caraco NF, McDowell WH, Tranvik LJ, Striegl RG, Duarte CM, Kortelainen P, Downing JA, Middelburg JJ, Melack J (2007) Plumbing the global carbon cycle: integrating inland waters into the terrestrial carbon budget. Ecosystems 10:171–184
Cook CDK, Urmikonig K (1984) A revision of the genus Ottelia (Hydrocharitaceae). 2. The species of Eurasia, Australasia and America. Aquat Bot 20:131–177
Cushman JC, Bohnert HJ (1999) Crassulacean acid metabolism: molecular genetics. Annu Rev Plant Physiol Plant Mol Biol 50:305–332
Degroote D, Kennedy RA (1977) Photosynthesis in Elodea canadensis Michx. 4 carbon acid synthesis. Plant Physiol 59:1133–1135
Edwards GE, Franceschi VR, Voznesenskaya EV (2004) Single-cell C4 photosynthesis versus the dual-cell (Kranz) paradigm. Annu Rev Plant Biol 55:173–196
Elzenga JTM, Prins HBA (1987) Light induced polarity of redox reactions in leaves of Elodea canadensis Michx. Plant Physiol 85:239–242
Farmer AM, Maberly SC, Bowes G (1986) Activities of carboxylation enzymes in freshwater macrophytes. J Exp Bot 37:1568–1573
Haimovich-Dayan M, Garfinkel N, Ewe D, Marcus Y, Gruber A, Wagner H, Kroth PG, Kaplan A (2013) The role of C4 metabolism in the marine diatom Phaeodactylum tricornutum. New Phytol 197:177–185
Hatch MD, Slack CR (1966) Photosynthesis by sugar-cane leaves. A new carboxylation reaction and the pathway of sugar formation. Biochem J 101:103–111
Holaday AS, Bowes G (1980) C4 acid metabolism and dark CO2 fixation in a submersed aquatic macrophyte (Hydrilla verticillata). Plant Physiol 65:331–335
Ibelings BW, Maberly SC (1998) Photoinhibition and the availability of inorganic carbon restrict photosynthesis by surface blooms of cyanobacteria. Limnol Oceanogr 43:408–419
Jansson M, Karlsson J, Jonsson A (2012) Carbon dioxide supersaturation promotes primary production in lakes. Ecol Lett 15:527–532
Kanai R, Edwards GE (1999) The biochemistry of C4 photosynthesis. In: Sage RF, Monson RK (eds) C4 plant biology. Academic Press, San Diego, pp 49–88
Keeley JE (1981) Isoetes howelli—a submerged aquatic CAM plant. Am J Bot 68:420–424
Keeley JE (1998) CAM photosynthesis in submerged aquatic plants. Bot Rev 64:121–175
Keeley JE, Sandquist DR (1992) Carbon: freshwater plants. Plant Cell Environ 15:1021–1035
Klavsen SK, Maberly SC (2009) Crassulacean acid metabolism contributes significantly to the in situ carbon budget in a population of the invasive aquatic macrophyte Crassula helmsii. Freshw Biol 54:105–118
Klavsen SK, Madsen TV, Maberly SC (2011) Crassulacean acid metabolism in the context of other carbon-concentrating mechanisms in freshwater plants: a review. Photosynth Res 109:269–279
Koch M, Bowes G, Ross C, Zhang X-H (2013) Climate change and ocean acidification effects on seagrasses and marine macroalgae. Glob Change Biol 19:103–132
Maberly SC (1996) Diel, episodic and seasonal changes in pH and concentrations of inorganic carbon in a productive lake. Freshw Biol 35:579–598
Maberly SC, Madsen TV (1998) Affinity for CO2 in relation to the ability of freshwater macrophytes to use HCO3. Funct Ecol 12:99–106
Maberly SC, Madsen TV (2002) Freshwater angiosperm carbon concentrating mechanisms: processes and patterns. Funct Plant Biol 29:393–405
Maberly SC, Spence DHN (1983) Photosynthetic inorganic carbon use by freshwater plants. J Ecol 71:705–724
Maberly SC, Barker PA, Stott AW, De Ville MM (2013) Catchment productivity control CO2 emissions from lakes. Nat Clim Change 3:391–394
Madsen TV (1987a) Interactions between internal and external CO2 pools in the photosynthesis of the aquatic CAM plants Littorella uniflora (L.) Aschers and Isoetes lacustris L. New Phytol 106:35–50
Madsen TV (1987b) Sources of inorganic carbon acquired through CAM in Littorella uniflora (L.) Aschers. J Exp Bot 38:367–377
Magnin NC, Cooley BA, Reiskind JB, Bowes G (1997) Regulation and localization of key enzymes during the induction of Kranz-less, C4-type photosynthesis in Hydrilla verticillata. Plant Physiol 115:1681–1689
Monson RK, Moore BD, Ku MSB, Edwards GE (1986) Co-function of C3-and C4-photosynthetic pathways in C3, C4 and C3–C4 intermediate Flaveria species. Planta 168:493–502
Moore B, Monson RK, Ku MSB, Edwards GE (1988) Activities of principal photosynthetic and photorespiratory enzymes in leaf mesophyll and bundle sheath protoplasts from the C3–C4 intermediate Flaveria ramosissima. Plant Cell Physiol 29:999–1006
Newman JR, Raven JA (1995) Photosynthetic carbon assimilation in Crassula helmsii. Oecologia 101:494–499
Ogren WL (2003) Affixing the O to Rubisco: discovering the source of photorespiratory glycolate and its regulation. Photosynth Res 76:53–63
Peisker M (1986) Models of carbon metabolism in C3–C4 intermediate plants as applied to the evolution of C4 photosynthesis. Plant Cell Environ 9:627–635
Raghavendra AS, Sage RF (2011) C4 Photosynthesis and Related CO2 Concentrating Mechanisms Introduction. In: Raghavendra AS, Sage RF (eds) C4 photosynthesis and related CO2 concentrating mechanisms, vol 32. Advances in photosynthesis and respiration. Springer, Dordrecht, pp 17–25
Raven JA (1970) Exogenous inorganic carbon sources in plant photosynthesis. Biol Rev 45:167–220
Raven JA, Giordano M, Beardall J, Maberly SC (2012) Algal evolution in relation to atmospheric CO2: carboxylases, carbon-concentrating mechanisms and carbon oxidation cycles. Philos Trans R Soc Lond B 367:493–507
Reinfelder JR (2011) Carbon concentrating mechanisms in eukaryotic marine phytoplankton. Annu Rev Mar Sci 3:291–315
Reinfelder JR, Kraepiel AML, Morel FMM (2000) Unicellular C4 photosynthesis in a marine diatom. Nature 407:996–999
Reiskind JB, Bowes G (1991) The role of phosphoenolpyruvate carboxykinase in a marine macroalga with C4-like photosynthetic characteristics. Proc Natl Acad Sci USA 88:2883–2887
Reiskind JB, Seamon PT, Bowes G (1988) Alternative methods of photosynthetic carbon assimilation in marine macroalgae. Plant Physiol 87:686–692
Reiskind JB, Madsen TV, VanGinkel LC, Bowes G (1997) Evidence that inducible C4-type photosynthesis is a chloroplastic CO2-concentrating mechanism in Hydrilla, a submersed monocot. Plant Cell Environ 20:211–220
Roberts K, Granum E, Leegood RC, Raven JA (2007) Carbon acquisition by diatoms. Photosynth Res 93:79–88
Sage RF, Christin P-A, Edwards EJ (2011) The C4 plant lineages of planet earth. J Exp Bot 62:3155–3169
Salvucci ME, Bowes G (1983) Two photosynthetic mechanisms mediating the low photorespiratory state in submersed aquatic angiosperms. Plant Physiol 73:488–496
Silvera K, Neubig KM, Whitten WM, Williams NH, Winter K, Cushman JC (2010) Evolution along the crassulacean acid metabolism continuum. Funct Plant Biol 37:995–1010
Talling JF (1985) Inorganic carbon reserves of natural waters and eco-physiological consequences of their photosynthetic depletion: microalgae. In: Lucas WJ, Berry JA (eds) Inorganic carbon uptake by photosynthetic organisms. The American Society of Plant Physiologists, Rockville, pp 404–420
Ueno O, Samejima M, Muto S, Miyachi S (1988) Photosynthetic characteristics of an amphibious plant, Elocharis vivipara. Expression of C4 and C3 modes in contrasting environments. Proc Natl Acad Sci USA 85:6733–6737
Von Caemmerer S (2000) Biochemical models of leaf photosynthesis, CSIRO, Collingwood, pp 123–140
Voznesenskaya EV, Franceschi VR, Kiirats O, Freitag H, Edwards GE (2001) Kranz anatomy is not essential for terrestrial C4 plant photosynthesis. Nature 414:543–546
Voznesenskaya EV, Franceschi VR, Kiirats O, Artyusheva EG, Freitag H, Edwards GE (2002) Proof of C4 photosynthesis without Kranz anatomy in Bienertia cycloptera (Chenopodiaceae). Plant J 31:649–662
Webb DR, Rattray MR, Brown JMA (1988) A preliminary survey for Crassulacean acid metabolism (CAM) in submerged aquatic macrophytes in New Zealand. N Z J Mar Freshw Res 22:231–235
Winter K, Foster JG, Edwards GE, Holtum JAM (1982) Intracellular-localization of enzymes of carbon metabolism in Mesembryanthemum crystallinum exhibiting C3 photosynthetic characteristics or performing Crassulacean acid metabolism. Plant Physiol 69:300–307
Xu J, Fan X, Zhang X, Xu D, Mou S, Cao S, Zheng Z, Miao J, Ye N (2012) Evidence of coexistence of C3 and C4 photosynthetic pathways in a green-tide-forming alga, Ulva prolifera. PLoS One 7:e37438
Acknowledgments
This research was partially supported by a Chinese Academy of Sciences Visiting Professorship for Senior International Scientists (2010T2S14, 2013T1S0021) and the National Scientific Foundation of China (30700083). Two anonymous referees helped us to improve the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, Y., Yin, L., Jiang, HS. et al. Biochemical and biophysical CO2 concentrating mechanisms in two species of freshwater macrophyte within the genus Ottelia (Hydrocharitaceae). Photosynth Res 121, 285–297 (2014). https://doi.org/10.1007/s11120-013-9950-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9950-y