Abstract
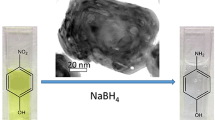
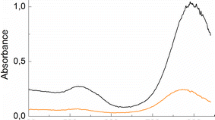
Gold nanostructures have been widely used as catalysts for chemical processes, energy conversion, and pollution control. The size of gold nanocatalysts is thus paramount for their catalytic activity. In this paper, gold nanorods with different sizes were prepared by means of the improved seeding growth approach by adding aromatic additive. The sizes and aspect ratios of the obtained gold nanorods were calculated according to the TEM characterization. Then, we studied the catalytic activities of gold nanorods using a model reaction based on the reduction of Eosin Y by NaBH4. By monitoring the absorption intensities of the radicals induced by gold nanorods in real time, we observed the clear size-dependent activity in the conversion of EY2− to EY3−. The conversion efficiency indicated that the gold nanorods with the smallest size were catalytically the most active probably due to their high number of coordinatively unsaturated surface atoms. In addition, a compensation effect dominated by the surface area of nanorods was observed in this catalytic reduction, which could be primarily attributed to the configuration of Eosin Y absorbed onto the surfaces of gold nanorods.









Similar content being viewed by others
References
Ahmadi TS, Wang ZL, Green TC, Henglein A, El-Sayed MA (1996) Shape-controlled synthesis of colloidal platinum nanoparticles. Science 272:1924–1926. doi:10.1126/science.272.5270.1924
Arvizo R, Bhattacharya R, Mukherjee P (2010) Gold nanoparticles: opportunities and challenges in nanomedicine. Expert Opin Drug Deliv 7:753–763. doi:10.1517/17425241003777010
Bannerjee NR, Negi AS (1973) Polarograms of eosin (2,4,5,7-tetrabromo-(R)-fluorescein) in aqueous buffers. Electrochim Acta 18:335–342. doi:10.1016/0013-4686(73)85001-7
Dreaden EC (2012) Chemistry, photophysics, and biomedical applications of gold nanotechnologies. Georgia Institute of Technology, Atlanta
Dreaden EC, Alkilany AM, Huang XH, Murphy CJ, El-Sayed MA (2012) The golden age: gold nanoparticles for biomedicine. Chem Soc Rev 41:2740–2779. doi:10.1039/C1CS15237H
Dreaden EC, Mackey MA, Huang X, Kang B, El-Sayed MA (2011) Beating cancer in multiple ways using nanogold. Chem Soc Rev 40:3391–3404. doi:10.1039/C0CS00180E
Fenger R, Fertitta E, Kirmse H, Thunemann AF, Rademann K (2012) Size dependent catalysis with CTAB-stabilized gold nanoparticles. Phys Chem Chem Phys 14:9343–9349. doi:10.1039/C2CP40792B
Ghosh P, Han G, De M, Kim CK, Rotello VM (2008) Gold nanoparticles in delivery applications. Adv Drug Delivery Rev 60:1307–1315. doi:10.1016/j.addr.2008.03.016
Giljohann DA, Seferos DS, Daniel WL, Massich MD, Patel PC, Mirkin CA (2010) Gold nanoparticles for biology and medicine. Angew Chem Int Ed 49:3280–3294. doi:10.1002/anie.200904359
Goux A, Pauporté T, Lincot D, Dunsch L (2007) In situ ESR and UV/Vis spectroelectrochemical study of Eosin Y upon reduction with and without Zn(II) Ions. ChemPhysChem 8:926–931. doi:10.1002/cphc.200700009
Han G, Ghosh P, Rotello VM (2007) Functionalized gold nanoparticles for drug delivery. Nanomedicine 2:113–123. doi:10.2217/17435889.2.1.113
Haruta M, Yamada N, Kobayashi T, Iijima S (1989) Gold catalysts prepared by coprecipitation for low-temperature oxidation of hydrogen and of carbon monoxide. J Catal 115:301–309. doi:10.1016/0021-9517(89)90034-1
Hutchings GJ (2005) Catalysis by gold. Catal Today 100:55–61. doi:10.1016/j.cattod.2004.12.016
Issa IM, Issa RM, Ghoneim MM, Temerk YM (1973) Polarography of eosin and erythrosin in solutions of varying pH at the DME. Electrochim Acta 18:265–270. doi:10.1016/0013-4686(73)80024-6
Kundu S, Lau S, Liang H (2009) Shape-controlled catalysis by cetyltrimethylammonium bromide terminated gold nanospheres, nanorods, and nanoprisms. J Phys Chem C 113:5150–5156. doi:10.1021/jp811331z
Li Y, Liu Q, Shen W (2011) Morphology-dependent nanocatalysis: metal particles. Dalton Trans 40:5811–5826. doi:10.1039/C0DT01404D
Lin F, Wise G (2009) Detection of proteins and sialoglycoproteins in polyacrylamide gels using eosin Y stain. In: Walker J (ed) The protein protocols handbook. Springer protocols handbooks. Humana Press, New York, pp 575–578. doi:10.1007/978-1-59745-198-7_53
Mahmoud MA, El-Sayed MA (2011) Time dependence and signs of the shift of the surface plasmon resonance frequency in nanocages elucidate the nanocatalysis mechanism in hollow nanoparticles. Nano Lett 11:946–953. doi:10.1021/nl103265s
Mahmoud MA, Weng GJ (2013) Nanocatalysis production of photoactive radicals. Catal Commun 38:63–66. doi:10.1016/j.catcom.2013.04.021
Mahmoud MA, Narayanan R, El-Sayed MA (2013) Enhancing colloidal metallic nanocatalysis: sharp edges and corners for solid nanoparticles and cage effect for hollow ones. Acc Chem Res. doi:10.1021/ar3002359
Mali SS, Betty CA, Bhosale PN, Patil PS (2012) Eosin-Y and N3-Dye sensitized solar cells (DSSCs) based on novel nanocoral TiO2: a comparative study. Electrochim Acta 59:113–120. doi:10.1016/j.electacta.2011.10.043
Nikoobakht B, El-Sayed MA (2003) Preparation and growth mechanism of gold nanorods (NRs) using seed-mediated growth method. Chem Mater 15:1957–1962. doi:10.1021/cm020732l
Orendorff CJ, Murphy CJ (2006) Quantitation of metal content in the silver-assisted growth of gold nanorods. J Phys Chem B 110:3990–3994. doi:10.1021/jp0570972
Panigrahi S et al (2007) Synthesis and size-selective catalysis by supported gold nanoparticles: study on heterogeneous and homogeneous catalytic process. J Phys Chem C 111:4596–4605. doi:10.1021/jp067554u
Saha K, Agasti SS, Kim C, Li X, Rotello VM (2012) Gold nanoparticles in chemical and biological sensing. Chem Rev 112:2739–2779. doi:10.1021/cr2001178
Sardar R, Funston AM, Mulvaney P, Murray RW (2009) Gold nanoparticles: past, present, and future. Langmuir 25:13840–13851. doi:10.1021/la9019475
Sau TK, Pal A, Pal T (2001) Size regime dependent catalysis by gold nanoparticles for the reduction of Eosin. J Phys Chem B 105:9266–9272. doi:10.1021/jp011420t
Sharma P, Brown S, Walter G, Santra S, Moudgil B (2006) Nanoparticles for bioimaging. Adv Colloid Interface Sci 123–126:471–485. doi:10.1016/j.cis.2006.05.026
Sperling RA, Rivera gil P, Zhang F, Zanella M, Parak WJ (2008) Biological applications of gold nanoparticles. Chem Soc Rev 37:1896–1908. doi:10.1039/B712170a
Srivastava S, Sharma SK, Sharma RK (2011) Synthesis of gold nanorods using concentrated aerosol OT in hexane and its application as catalyst for the reduction of eosin. Colloids Surf A 373:61–65. doi:10.1016/j.colsurfa.2010.10.022
Thompson DT (2007) Using gold nanoparticles for catalysis. Nano Today 2:40–43. doi:10.1016/s1748-0132(07)70116-0
Weng GJ, Mahmoud MA, El-Sayed MA (2012) Nanocatalysts can change the number of electrons involved in oxidation–reduction reaction with the nanocages being the most efficient. J Phys Chem C 116:24171–24176. doi:10.1021/jp308869m
Wilson R (2008) The use of gold nanoparticles in diagnostics and detection. Chem Soc Rev 37:2028–2045. doi:10.1039/B712179m
Xia L, Zhao H, Liu G, Hu X, Liu Y, Li J, Yang D (2011) Degradation of dyes using hollow copper microspheres as catalyst. Colloids Surf A 384:358–362. doi:10.1016/j.colsurfa.2011.04.016
Ye X, Jin L, Caglayan H, Chen J, Xing G, Zheng C, Doan-Nguyen V, Kang Y, Engheta N, Kagan CR, Murray CB (2012) Improved size-tunable synthesis of monodisperse gold nanorods through the use of aromatic additives. ACS Nano 6:2804–2817. doi:10.1021/nn300315j
Ye X, Zheng C, Chen J, Gao Y, Murray CB (2013) Using binary surfactant mixtures to simultaneously improve the dimensional tunability and monodispersity in the seeded growth of gold nanorods. Nano Lett 13:765–771. doi:10.1021/nl304478h
Yoshida T et al (2009) Electrodeposition of inorganic/organic hybrid thin films. Adv Funct Mater 19:17–43. doi:10.1002/adfm.200700188
Zeng J, Zhang Q, Chen JY, Xia YN (2010) A comparison study of the catalytic properties of Au-based nanocages, nanoboxes, and nanoparticles. Nano Lett 10:30–35. doi:10.1021/nl903062e
Zhang JB, Sun LN, Yoshida T (2011) Spectroelectrochemical studies on redox reactions of eosin Y and its polymerization with Zn2+ ions. J Electroanal Chem 662:384–395. doi:10.1016/j.jelechem.2011.09.008
Zhou X, Xu W, Liu G, Panda D, Chen P (2009) Size-dependent catalytic activity and dynamics of gold nanoparticles at the single-molecule level. J Am Chem Soc 132:138–146. doi:10.1021/ja904307n
Zhu Y, Qian H, Das A, Jin R (2011) Comparison of the catalytic properties of 25-atom gold nanospheres and nanorods. Chin J Catal 32:1149–1155. doi:10.1016/S1872-2067(10)60238-0
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant Nos. 21403161 and 61178075) and the China Post-doctoral Science Foundation (Grant No. 2014M552433). G.W. acknowledges the support from Xi’an Jiaotong University Faculty Research Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weng, G., Qi, Y., Li, J. et al. Size-dependent production of radicals in catalyzed reduction of Eosin Y using gold nanorods. J Nanopart Res 17, 367 (2015). https://doi.org/10.1007/s11051-015-3175-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-015-3175-2