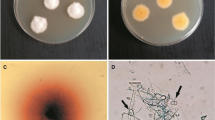
Abstract
A new black yeast species, Exophiala xenobiotica, is described, a segregant of the Exophiala jeanselmei complex. It is morphologically very similar to E. jeanselmei, though with less melanized conidiogenous cells, but deviates unambiguously on the basis of molecular phylogeny. The species is a relatively common agent of cutaneous infections in humans, whereas E. jeanselmei is associated with subcutaneous infections. Environmental strains of E. xenobiotica are frequently found in habitats rich in monoaromatic hydrocarbons and alkanes.
Similar content being viewed by others
References
Arias M, Stotzky G (1997) Adsorption and binding of copper and lead by Exophiala sp. Abstr Gen Meet ASM 97:497
Cox HHJ, Moerman RE, Van Baalen S, Van Heyningen WJM, Doddema HJ, Harder W (1997) Performance of a styrene-degrading biofilter containing the yeast Exophiala jeanselmei. Biotechnol Bioeng 53:259–266
De Hoog GS, Guarro J, Gené J, Figueras MJ (2000) Atlas of Clinical Fungi, 2nd ed. Centraalbureau voor Schimmelcultures/Universitat Rovira i Virgili, Utrecht/Reus
De Hoog GS, Vicente V, Caligiorne RB, Kantargliocu S, Tintelnot K, Gerrits van den Ende AHG, Haase G (2003) Species diversity and polymorphism in the Exophiala spinifera clade containing opportunistic black yeast-like fungi. J Clin Microbiol 41:4767–4778
De Hoog GS (1977) Rhinocladiella and allied genera. Stud Mycol 15:141–144
De Rijk P, De Wachter R (1993) DCSE v. 2.54, an interactive tool for sequence alignment and secondary structure research. Comput Appl Biosci 9:735–740
Elborn JS (1999) Treatment of Staphylococcus aureus in cystic fibrosis. Thorax 54:377–378
Engemann J, Kaye K, Cox G, Perfect J, Schell W,␣McGarry SA, Patterson K, Edupuganti S, Cook P, Rutala WA, Weber DJ, Hoffmann KK, Engel J, Young S, Durant E, McKinnon K, Cobb N, Bell L,␣Gibson J, Jernigan D, Arduino M, Fridkin S, Archibald L, Sehulster L, Morgan J, Hajjeh R, Brandt M, Warnock D, Duffus WA (2002) Exophiala infection from contaminated injectable steroids prepared by a compounding pharmacy. Center for Disease Control (CDC) Morbid Mortal Wkly Rep 51:1109–1112
Göttlich E, Van der Lubbe W, Lange B, Fiedler S, Melchert I, Reifenrath M, Flemming H-C, De Hoog GS (2002) Fungal flora in groundwater-derived public drinking water. Int J Hyg Environ Health 205:269–279
Haase G, Sonntag L, Melzer-Krick B, De Hoog GS (1999) Phylogenetic inference by SSU-gene analysis of members of the Herpotrichiellaceae with special reference to human pathogenic species. Stud Mycol 43:80–97
Hölker U, Bend J, Pracht R, Müller T, Tetsch L, De Hoog GS (2003) Hortaea acidophila, a new acidophilic black yeast from lignite. Antonie van Leeuwenhoek 86: 287–294
Iwatsu T, Miyaji M, Okamoto S (1981) Isolation of Phialophora verrucosa and Fonsecaea pedrosoi from nature in Japan. Mycopathologia 75: 149–158.
Iwatsu T, Nishimura K, Miyaji M (1984) Exophiala castellanii sp. nov. Mycotaxon 20:307–314
Kawasaki M, Ishizaki H, Matsumoto T, Matsuda T, Nishimura K, Miyaji M (1999) Mitochondrial DNA analysis of Exophiala jeanselmei var. lecanii-corni and Exophiala castellanii. Mycopathologia 146:75–77
Langeron M (1928) Mycétome à Torula jeanselmei Langeron, 1928. Nouveau type de mycétome à grains noirs. Ann Parasitol Hum Comp 6:385–403
Ludwig W, Strunk O, Westram R, Richter L, Meier H, Yadhukumar, Buchner A, Lai T, Steppi S, Jobb G, Forster W, Brettske I, Gerber S, Ginhart AW, Gross␣O, Grumann S, Hermann S, Jost R, Konig A, Liss T, Lussmann R, May M, Nonhoff B, Reichel B, Strehlow R, Stamatakis A, Stuckmann N, Vilbig A, Lenke M, Ludwig T, Bode A, Schleifer KH (2004) ARB: a software environment for sequence data. Nucleic Acids Res 32: 1363–1371.
Matos T, De Hoog GS, De Boer AG, De Crom I, Haase G (2002) High prevalence of the neurotrope Exophiala dermatitidis and related oligotrophic black yeasts in sauna facilities. Mycoses 45:373–377
Matsuda M, Naka W, Tajima S, Harada T, Nishikawa T, Kaufman L, Standard P (1989) Deoxyribonucleic acid hybridization studies of Exophiala dermatitidis and Exophiala jeanselmei. Microbiol Immunol 33:631–639
McGinnis MR, Padhye AA (1977) Exophiala jeanselmei, a new combination for Phialophora jeanselmei. Mycotaxon 5:341–352
Middelhoven WJ (1993) Catabolism of benzene compounds by ascomycetous and basidiomycetous yeasts and yeastlike fungi. Antonie van Leeuwenhoek 63:125–144
Middelhoven WJ, De Hoog GS, Notermans C (1989) Carbon assimilation and extracellular antigens of some yeast-like fungi. Antonie van Leeuwenhoek 55:165–175
Murray IG, Dunkerley GE, Hughes KEA (1963) A case of Madura foot caused by Phialophora jeanselmei. Sabouraudia 3:175–177
National National Committee for Clinical Laboratory Standards (2002) Reference method for broth dilution antifungal susceptibility testing of conidium forming filamentous fungi. Proposed standard M38-A. National Committee for Clinical Laboratory Standards, Wayne, Pa.
Neumeister B, Zollner TM, Krieger D, Sterry W, Marre R (1995) Mycetoma due to Exophiala jeanselmei and Mycobacterium chelonae in a 73-year-old man with idiopathic CD4+ T lymphocytopenia. Mycoses 38:271–276
Nucci M, Akiti T, Barreiros G, Silveira F, Revankar SG, Wickes BL, Sutton DA, Patterson TF (2002) Nosocomial outbreak of Exophiala jeanselmei fungemia associated with contamination of hospital water. Clin Infect Dis 34:1475–1480
Page RDM (1996) TREEVIEW: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Phillips G, McEwan H, McKay I, Crowe G, McBeath J (1998) Black pigmented fungi in the water pipe-work supplying endoscope washer disinfectors. J Hosp Infect 40:250–251
Porteous NB, Redding SW, Thompson EH, Grooters AM, De Hoog GS, Sutton DA (2003) The isolation of an unusual fungus in treated dental unit waterlines. J Am Dent Assoc 134:467–476
Prenafeta-Boldú FX, Kuhn A, Luykx DMAM, Anke H, Van Groenestijn JW, De Bont J (2001) Isolation and characterisation of fungi growing on volatile aromatic hydrocarbons as their sole carbon and energy source. Mycol Res 105:477–484
Prenafeta-Boldú FX, Summerbell R, De Hoog GS (2006) Fungi growing on aromatic hydrocarbons: biotechnology’s unexpected encounter with biohazard. FEMS Microbiol Rev 30:109–130
Sterflinger K, Prillinger H (2001) Molecular taxonomy and biodiversity of rock fungal communities in an urban environment (Vienna, Austria). Antonie van Leeuwenhoek 80:275–286
Sutton DA, Fothergill AW, Rinaldi MG (1998) Guide to clinically significant fungi. Williams & Wilkins, Baltimore
Sutton DA, Sanche SE, Revankar SG, Fothergill AW, Rinaldi MG (1999a) In vitro amphotericin B resistance in clinical isolates of Aspergillus terreus, with a head-to-head comparison to voriconazole. J Clin Microbiol 37:2343–2345
Swofford DL (2000) PAUP* 4.0: phylogenetic analysis using parsimony. Sinauer Associates, Sunderland, MA, USA
Untereiner WA (1994) A simple method for the in vitro production of pseudothecia in species of Capronia. Mycologia 86:290–295
Van de Peer Y, De Wachter R (1994) Treecon for Windows: a software package for the construction and drawing of evolutionary trees for the Microsoft Windows environment. Comput Appl Biosci 10:569–570
Vitale RG, De Hoog GS (2002) Molecular diversity, new species and antifungal susceptibilities in the Exophiala spinifera clade. Med Mycol 40:545–556
Wang L, Yokoyama K, Miyaji M, Nishimura K (2001) Identification, classification and phylogeny of the pathogenic species Exophiala jeanselmeiand related species by mitochondrial cytochrome b gene analysis. J Clin Microbiol 39:4462–4467
Watson PR, Sanford PA, Burton KA, Cadmus MC, Jeanes A (1976) An extracellular fungal polysaccharide composed of 2-acetamido-2-deoxy-d-glucuronic acid residues. Carbohydr Res 46:259–265
Woollons A, Darley CR, Pandian S, Blackee J, Paul J (1996) Phaeohyphomycosis caused by Exophiala dermatitidis following intra-articular steroid injection. Br J Dermatol 135:475–477
Zeng JS, Sutton DA, De Hoog GS (2006) Identification and antifungal susceptibility of clinical isolates of the genus Exophiala from the USA (submitted)
Acknowledgements
K. Sterflinger (Vienna, Austria), U. Hölker (Bonn, Germany), L. Sigler (Edmonton, Canada), G. Haase (Aachen, Germany) and the curator of the IFM culture collection (Chiba, Japan) are acknowledged for sending strains. A. H. G. Gerrits van den Ende and K. F. Luijsterburg are thanked for technical assistance, and R.C. Summerbell for comments on the text.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Hoog, G.S., Zeng, J.S., Harrak, M.J. et al. Exophiala xenobiotica sp. nov., an opportunistic black yeast inhabiting environments rich in hydrocarbons. Antonie van Leeuwenhoek 90, 257–268 (2006). https://doi.org/10.1007/s10482-006-9080-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-006-9080-z