Abstract
Background
The localization and role of the calcium-sensing receptor (CaSR) along the nephron including the collecting ducts is still open to debate.
Methods
Using the quantitative, highly sensitive in situ hybridization technique and a double-staining immunohistochemistry technique, we investigated the axial distribution and expression of CaSR along the nephron in mice (C57B/6J) treated for 6 days with acid or alkali diets.
Results
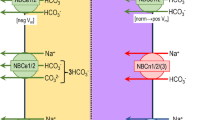
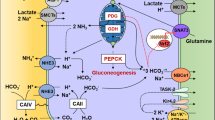
Under control condition, CaSR was specifically localized in the cortical and medullary thick ascending limb of Henle’s loop (CTAL and MTAL), macula densa (MD), distal convoluted tubule (DCT), and CCD (TALs, MD > DCT, CCD). Along the CCD, CaSR was co-localized with an anion exchanger type 4 (AE4), a marker of the basolateral membrane of type-B intercalated cell (IC-B) in mice. On the contrary, CaSR was not detected either in principal cells (PC) or in type-A intercalated cell (IC-A). CaSR expression levels in IC-B significantly (P < 0.005) decreased when mice were fed NH4Cl (acid) diets and increased when animals were given NaHCO3 (alkali) diets. As expected, cell heights of IC-A and IC-B significantly (P < 0.005) increased in the above experimental conditions. Surprisingly, single infusion (ip) of neomycin, an agonist of CaSR, significantly (P < 0.005) increased urinary Ca excretion without further increasing the hourly urine volume and significantly (P < 0.05) decreased urine pH.
Conclusion
CaSR, cloned from rat kidney, was localized in the basolateral membrane of IC-B and was more expressed during alkali-loading. Its alkali-sensitive expression may promote urinary alkali secretion for body acid–base balance.









Similar content being viewed by others
References
Fenton RA, Knepper MA. Mouse models and the urinary concentrating mechanism in the new millennium. Physiol Rev. 2007;87:1083–112.
Wang WH, Giebisch G. Regulation of potassium (K) handling in the renal collecting duct. Pflügers Arch. 2009;458:157–68.
Staruschenko A. Regulation of transport in the connecting tubule and cortical collecting duct. Compr Physiol. 2012;2:1541–84.
Hebert SC, Brown EM, Harris HW. Role of the Ca2+-sensing receptor in divalent mineral ion homeostasis. J Exp Biol. 1997;200:295–302.
Brown EM, Gamba G, Riccardi D, Lombardi M, Butters R, Kifor O, Sun A, Hediger MA, Lytton J, Hebert SC. Cloning and characterization of an extracellular Ca2+-sensing receptor from bovine parathyroid. Nature. 1993;366:575–80.
Brown EM, Pollak M, Riccardi D, Hebert SC. Cloning and characterization of an extracellular Ca2+-sensing receptor from parathyroid and kidney: new insights into the physiology and pathophysiology of calcium metabolism. Nephrol Dial Transplant. 1994;9:1703–6.
Houillier P. Calcium-sensing in the kidney. Curr Opin Nephrol Hypertens. 2013;22:566–71.
Alfadda TI, Saleh AM, Houillier P, Geibel JP. Calcium-sensing receptor 20 years later. Am J Physiol Cell Physiol. 2014;307:C221–31.
Riccardi D, Park J, Lee WS, Gamba G, Brown EM, Hebert SC. Cloning and functional expression of a rat kidney extracellular calcium/polyvalent cation-sensing receptor. Proc Natl Acad Sci USA. 1995;92:131–5.
Riccardi D, Lee WS, Lee K, Segre GV, Brown EM, Hebert SC. Localization of the extracellular Ca2+-sensing receptor and PTH/PTHrP receptor in rat kidney. Am J Physiol. 1996;271:F951–6.
Chattopadhyay N, Baum M, Bai M, Riccardi D, Hebert SC, Harris HW, Brown EM. Ontogeny of the extracellular calcium-sensing receptor in rat kidney. Am J Physiol. 1996;271:F736–43.
Sands JM, Naruse M, Baum M, Jo I, Hebert SC, Brown EM, Harris HW. Apical extracellular calcium/polyvalent cation-sensing receptor regulates vasopressin-elicited water permeability in rat kidney inner medullary collecting duct. J Clin Invest. 1997;99:1399–405.
Riccardi D, Hall AE, Chattopadhyay N, Xu JZ, Brown EM, Hebert SC. Localization of the extracellular Ca2+/polyvalent cation-sensing protein in rat kidney. Am J Physiol. 1998;274:F611–22.
Yang T, Hassan S, Huang YG, Smart AM, Briggs JP, Schnermann JB. Expression of PTHrP, PTH/PTHrP receptor, and Ca2+-sensing receptor mRNAs along the rat nephron. Am J Physiol. 1997;272:F751–8.
Loupy A, Ramakrishnan SK, Wootla B, Chambrey R, de la Faille R, Bourgeois S, Bruneval P, Mandet C, Christensen EI, Faure H, Cheval L, Laghmani K, Collet C, Eladari D, Dodd RH, Ruat M, Houillier P. PTH-independent regulation of blood calcium concentration by the calcium-sensing receptor. J Clin Invest. 2012;122:3355–67.
Toka HR, Al-Romaih K, Koshy JM, DiBartolo S 3rd, Kos CH, Quinn SJ, Curhan GC, Mount DB, Brown EM, Pollak MR. Deficiency of the calcium-sensing receptor in the kidney causes parathyroid hormone-independent hypocalciuria. J Am Soc Nephrol. 2012;23:1879–90.
Casare F, Milan D, Fernandez R. Stimulation of calcium-sensing receptor increases biochemical H+-ATPase activity in mouse cortex and outer medullary regions. Can J Physiol Pharmacol. 2014;92:181–8.
Kerstens HM, Poddighe PJ, Hanselaar AG. A novel in situ hybridization signal amplification method based on the deposition of biotinylated tyramine. Histochem Cytochem. 1995;43:347–52.
Suzuki T, Kadoya Y, Sato Y, Handa K, Takahashi T, Kakita A, Yamashina S. The expression of pancreatic endocrine markers in centroacinar cells of the normal and regenerating rat pancreas: their possible transformation to endocrine cells. Arch Histol Cytol. 2003;66:347–58.
Kobayashi M, Yasuoka Y, Sato Y, Zhou M, Abe H, Kawahara K, Okamoto H. Upregulation of calbindin D28k in the late distal tubules in the potassium-loaded adrenalectomized mouse kidney. Clin Exp Nephrol. 2011;15:355–62.
Kriz W, Bankir L. A standard nomenclature for structures of the kidney. Kidney Int. 1988;33:1–7.
Madsen KM, Tisher CC. Response of intercalated cells of rat outer medullary collecting duct to chronic metabolic acidosis. Lab Invest. 1984;51:268–76.
Quinn SJ, Bai M, Brown EM. pH sensing by the calcium-sensing receptor. J Biol Chem. 2004;279:37241–9.
Ikebe M, Nonoguchi H, Nakayama Y, Tashima Y, Tomita K. Upregulation of the secretory-type Na+/K+/2Cl−-cotransporter in the kidney by metabolic acidosis and dehydration in rats. J Am Soc Nephrol. 2001;12:423–30.
Wagner CA, Finberg KE, Stehberger PA, Lifton RP, Giebisch GH, Aronson PS, Geibel JP. Regulation of the expression of the Cl−/anion exchanger pendrin in mouse kidney by acid-base status. Kidney Int. 2002;62:2109–17.
Yasuoka Y, Kobayashi M, Sato Y, Nonoguchi H, Tanoue A, Okamoto H, Kawahara K. Decreased expression of aquaporin 2 in the collecting duct of mice lacking the vasopressin V1a receptor. Clin Exp Nephrol. 2013;17:183–90.
Sumida K, Nakamura M, Ubara Y, Marui Y, Tanaka K, Takaichi K, Tomikawa S, Inoshita N, Ohashi K. Cinacalcet upregulates calcium-sensing receptors of parathyroid glands in hemodialysis patients. Am J Nephrol. 2013;37:405–12.
Blomqvist SR, Vidarsson H, Fitzgerald S, Johansson BR, Ollerstam A, Brown R, Persson AEG, Bergström G, Enerbäck S. Distal renal tubular acidosis in mice that lack the forkhead transcription factor Foxi1. J Clin Invest. 2004;113:1560–70.
Yasuoka Y, Kobayashi M, Sato Y, Zhou M, Abe H, Okamoto H, Nonoguchi H, Tanoue A, Kawahara K. The intercalated cells of the mouse kidney OMCDis are the target of the vasopressin V1a receptor axis for urinary acidification. Clin Exp Nephrol. 2013;17:783–92.
Wall SM, Hassell KA, Royaux IE, Green ED, Chang JY, Shipley GL, Verlander JW. Localization of pendrin in mouse kidney. Am J Physiol Renal Physiol. 2003;284:F229–41.
Chambrey R, Kurth I, Peti-Peterdi J, Houillier P, Purkerson JM, Leviel F, Hentschke M, Zdebik AA, Schwartz GJ, Hübner CA, Eladari D. Renal intercalated cells are rather energized by a proton than a sodium pump. Proc Natl Acad Sci USA. 2013;110:7928–33.
Purkerson JM, Heintz EV, Nakamori A, Schwartz GJ. Insights into acidosis-induced regulation of SLC26A4 (pendrin) and SLC4A9 (AE4) transporters using three-dimensional morphometric analysis of β-intercalated cells. Am J Physiol Renal Physiol. 2014;307:F601–11.
Tsuganezawa H, Kobayashi K, Iyori M, Araki T, Koizumi A, Watanabe S, Kaneko A, Fukao T, Monkawa T, Yoshida T, Kim DK, Kanai Y, Endou H, Hayashi M, Saruta T. A new member of the HCO3 − transporter superfamily is an apical anion exchanger of β-intercalated cells in the kidney. J Biol Chem. 2001;276:8180–9.
Ko SB, Luo X, Hager H, Rojek A, Choi JY, Licht C, Suzuki M, Muallem S, Nielsen S, Ishibashi K. AE4 is a DIDS-sensitive Cl−/HCO3 − exchanger in the basolateral membrane of the renal CCD and the SMG duct. Am J Physiol Cell Physiol. 2002;283:C1206–18.
Xu J, Barone S, Petrovic S, Wang Z, Seidler U, Riederer B, Ramaswamy K, Dudeja PK, Shull GE, Soleimani M. Identification of an apical Cl−/HCO3 − exchanger in gastric surface mucous and duodenal villus cells. Am J Physiol Gastrointest Liver Physiol. 2003;285:G1225–34.
Loretz CA. Extracellular calcium-sensing receptors in fishes. Comp Biochem Physiol A Mol Integr Physiol. 2008;149:225–45.
Brown EM, MacLeod RJ. Extracellular calcium sensing and extracellular calcium signaling. Physiol Rev. 2001;81:239–97.
Riccardi D, Brown EM. Physiology and pathophysiology of the calcium-sensing receptor in the kidney. Am J Physiol Renal Physiol. 2010;298:F485–99.
Renkema KY, Velic A, Dijkman HB, Verkaart S, van der Kemp AW, Nowik M, Timmermans K, Doucet A, Wagner CA, Bindels RJ, Hoenderop JG. The calcium-sensing receptor promotes urinary acidification to prevent nephrolithiasis. J Am Soc Nephrol. 2009;20:1705–13.
Bergsland KJ, Coe FL, Gillen DL, Worcester EM. A test of the hypothesis that the collecting duct calcium-sensing receptor limits rise of urine calcium molarity in hypercalciuric calcium kidney stone formers. Am J Physiol Renal Physiol. 2009;297:F1017–23.
Brown D, Wagner CA. Molecular mechanisms of acid-base sensing by the kidney. J Am Soc Nephrol. 2012;23:774–80.
Acknowledgments
This study was supported by grants from the Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan to Y.Y. (No. 24790222) and to K.K. (No. 23591224, 26461259). Special thanks are given to Y.Nakabayashi (Kitasato Univ Sch of Med) for her technical assistance. Parts of this study were presented at the meetings of American Society of Nephrology (Philadelphia, 2011) and International Union of Physiological Sciences (Birmingham (UK), 2013).
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yasuoka, Y., Sato, Y., Healy, J.M. et al. pH-sensitive expression of calcium-sensing receptor (CaSR) in type-B intercalated cells of the cortical collecting ducts (CCD) in mouse kidney. Clin Exp Nephrol 19, 771–782 (2015). https://doi.org/10.1007/s10157-014-1063-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-1063-1