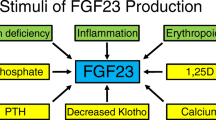
Abstract
Fibroblast growth factor 23 (FGF23) is an “endocrine” FGF acting in the kidney as a phosphaturic hormone and a suppressor of active vitamin D, through an inhibition of the 1α hydroxylase and a stimulation of the 24 hydroxylase. Beyond its well-known effects on the bone/kidney/parathyroid axis and its deregulation during chronic kidney disease (CKD), recent evidence has revealed its direct systemic effects on cardiovascular health. In the meantime, studies have highlighted the health implications for vitamin D inside and outside CKD that also extend beyond its classical actions on mineral homeostasis and bone metabolism: vitamin D has indeed been shown to exert pluripotent non-classical effects as a modulator of immune function in monocytes, mainly through the stimulation of the antimicrobial cathelicidin. The aim of this review is to provide new insights on the interplay between FGF23 and vitamin D in innate immunity in the context of CKD.
Similar content being viewed by others
References
Liu S, Quarles LD (2007) How fibroblast growth factor 23 works. J Am Soc Nephrol 18:1637–1647
Yamazaki Y, Tamada T, Kasai N, Urakawa I, Aono Y, Hasegawa H, Fujita T, Kuroki R, Yamashita T, Fukumoto S, Shimada T (2008) Anti-FGF23 neutralizing antibodies show the physiological role and structural features of FGF23. J Bone Miner Res 23:1509–1518
Yoshiko Y, Wang H, Minamizaki T, Ijuin C, Yamamoto R, Suemune S, Kozai K, Tanne K, Aubin JE, Maeda N (2007) Mineralized tissue cells are a principal source of FGF23. Bone 40:1565–1573
Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, Fujita T, Nakahara K, Fukumoto S, Yamashita T (2004) FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res 19:429–435
Razzaque MS (2009) Does FGF23 toxicity influence the outcome of chronic kidney disease? Nephrol Dial Transplant 24:4–7
Krajisnik T, Bjorklund P, Marsell R, Ljunggren O, Akerstrom G, Jonsson KB, Westin G, Larsson TE (2007) Fibroblast growth factor-23 regulates parathyroid hormone and 1alpha-hydroxylase expression in cultured bovine parathyroid cells. J Endocrinol 195:125–131
Ben-Dov IZ, Galitzer H, Lavi-Moshayoff V, Goetz R, Kuro-o M, Mohammadi M, Sirkis R, Naveh-Many T, Silver J (2007) The parathyroid is a target organ for FGF23 in rats. J Clin Invest 117:4003–4008
Shimada T, Urakawa I, Isakova T, Yamazaki Y, Epstein M, Wesseling-Perry K, Wolf M, Salusky IB, Juppner H (2010) Circulating fibroblast growth factor 23 in patients with end-stage renal disease treated by peritoneal dialysis is intact and biologically active. J Clin Endocrinol Metab 95:578–585
Lafage-Proust MH (2010) Does the downregulation of the FGF23 signaling pathway in hyperplastic parathyroid glands contribute to refractory secondary hyperparathyroidism in CKD patients? Kidney Int 77:390–392
Juppner H, Wolf M, Salusky IB (2010) FGF23: more than a regulator of renal phosphate handling? J Bone Miner Res 25:2091–2097
Prie D, Urena Torres P, Friedlander G (2009) Latest findings in phosphate homeostasis. Kidney Int 75:882–889
Isakova T, Xie H, Yang W, Xie D, Anderson AH, Scialla J, Wahl P, Gutierrez OM, Steigerwalt S, He J, Schwartz S, Lo J, Ojo A, Sondheimer J, Hsu CY, Lash J, Leonard M, Kusek JW, Feldman HI, Wolf M (2011) Fibroblast growth factor 23 and risks of mortality and end-stage renal disease in patients with chronic kidney disease. JAMA 305:2432–2439
Mirza MA, Hansen T, Johansson L, Ahlstrom H, Larsson A, Lind L, Larsson TE (2009) Relationship between circulating FGF23 and total body atherosclerosis in the community. Nephrol Dial Transplant 24:3125–3131
Mirza MA, Larsson A, Lind L, Larsson TE (2009) Circulating fibroblast growth factor-23 is associated with vascular dysfunction in the community. Atherosclerosis 205:385–390
Fukagawa M, Nii-Kono T, Kazama JJ (2005) Role of fibroblast growth factor 23 in health and in chronic kidney disease. Curr Opin Nephrol Hypertens 14:325–329
Gutierrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, Shah A, Smith K, Lee H, Thadhani R, Juppner H, Wolf M (2008) Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med 359:584–592
Nakanishi S, Kazama JJ, Nii-Kono T, Omori K, Yamashita T, Fukumoto S, Gejyo F, Shigematsu T, Fukagawa M (2005) Serum fibroblast growth factor-23 levels predict the future refractory hyperparathyroidism in dialysis patients. Kidney Int 67:1171–1178
Srivaths PR, Goldstein SL, Silverstein DM, Krishnamurthy R, Brewer ED (2011) Elevated FGF 23 and phosphorus are associated with coronary calcification in hemodialysis patients. Pediatr Nephrol 26:945–951
Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, Gutierrez OM, Aguillon-Prada R, Lincoln J, Hare JM, Mundel P, Morales A, Scialla J, Fischer M, Soliman EZ, Chen J, Go AS, Rosas SE, Nessel L, Townsend RR, Feldman HI, St John Sutton M, Ojo A, Gadegbeku C, Di Marco GS, Reuter S, Kentrup D, Tiemann K, Brand M, Hill JA, Moe OW, Kuro OM, Kusek JW, Keane MG, Wolf M (2011) FGF23 induces left ventricular hypertrophy. J Clin Invest 121:4393–4408
Chonchol M, Kendrick J, Targher G (2011) Extra-skeletal effects of vitamin D deficiency in chronic kidney disease. Ann Med 43:273–282
Adams JS, Hewison M (2010) Update in vitamin D. J Clin Endocrinol Metab 95:471–478
Adams JS, Hewison M (2008) Unexpected actions of vitamin D: new perspectives on the regulation of innate and adaptive immunity. Nat Clin Pract Endocrinol Metab 4:80–90
Bacchetta J, Ranchin B, Dubourg L, Cochat P (2010) Vitamin D revisited: a cornerstone of health? Arch Pediatr 17:1687–1695
Liu PT, Stenger S, Li H, Wenzel L, Tan BH, Krutzik SR, Ochoa MT, Schauber J, Wu K, Meinken C, Kamen DL, Wagner M, Bals R, Steinmeyer A, Zugel U, Gallo RL, Eisenberg D, Hewison M, Hollis BW, Adams JS, Bloom BR, Modlin RL (2006) Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 311:1770–1773
Adams JS, Ren S, Liu PT, Chun RF, Lagishetty V, Gombart AF, Borregaard N, Modlin RL, Hewison M (2009) Vitamin D-directed rheostatic regulation of monocyte antibacterial responses. J Immunol 182:4289–4295
Menon S, Valentini RP, Hidalgo G, Peschansky L, Mattoo TK (2008) Vitamin D insufficiency and hyperparathyroidism in children with chronic kidney disease. Pediatr Nephrol 23:1831–1836
Belostotsky V, Mughal MZ, Berry JL, Webb NJ (2008) Vitamin D deficiency in children with renal disease. Arch Dis Child 93:959–962
Bacchetta J, Dubourg L, Harambat J, Ranchin B, Abou-Jaoude P, Arnaud S, Carlier MC, Richard M, Cochat P (2010) The Influence of Glomerular Filtration Rate and Age on Fibroblast Growth Factor 23 Serum Levels in Pediatric Chronic Kidney Disease. J Clin Endocrinol Metab 95:1741–1748
Shroff R, Wan M, Gullet A, Ledermann S, Shute R, Knott C, Wells D, Aitkenhead H, Manickavasagar B, van’t Hoff W, Rees L (2012) Ergocalciferol supplementation in children with CKD delays the onset of secondary hyperparathyroidism: a randomized trial. Clin J Am Soc Nephrol doi:10.2215/CJN.04760511
Parasuraman R, Samarapungavan D, Venkat KK (2010) Updated principles and clinical caveats in the management of infection in renal transplant recipients. Transplant Rev (Orlando) 24:43–51
Cohen G, Haag-Weber M, Horl WH (1997) Immune dysfunction in uremia. Kidney Int Suppl 62:S79–S82
Dalrymple LS, Go AS (2008) Epidemiology of acute infections among patients with chronic kidney disease. Clin J Am Soc Nephrol 3:1487–1493
Viasus D, Garcia-Vidal C, Cruzado JM, Adamuz J, Verdaguer R, Manresa F, Dorca J, Gudiol F, Carratala J (2011) Epidemiology, clinical features and outcomes of pneumonia in patients with chronic kidney disease. Nephrol Dial Transplant 26:2899–2906
Sarnak MJ, Jaber BL (2001) Pulmonary infectious mortality among patients with end-stage renal disease. Chest 120:1883–1887
Sarnak MJ, Jaber BL (2000) Mortality caused by sepsis in patients with end-stage renal disease compared with the general population. Kidney Int 58:1758–1764
Allon M, Depner TA, Radeva M, Bailey J, Beddhu S, Butterly D, Coyne DW, Gassman JJ, Kaufman AM, Kaysen GA, Lewis JA, Schwab SJ (2003) Impact of dialysis dose and membrane on infection-related hospitalization and death: results of the HEMO Study. JASN 14:1863–1870
Harambat J, van Stralen KJ, Kim JJ, Tizard EJ (2012) Epidemiology of chronic kidney disease in children. Pediatr Nephrol 27:363–373
Mong Hiep TT, Ismaili K, Collart F, Van Damme-Lombaerts R, Godefroid N, Ghuysen MS, Van Hoeck K, Raes A, Janssen F, Robert A (2010) Clinical characteristics and outcomes of children with stage 3–5 chronic kidney disease. Pediatr Nephrol 25:935–940
Sardenberg C, Suassuna P, Andreoli MC, Watanabe R, Dalboni MA, Manfredi SR, dos Santos OP, Kallas EG, Draibe SA, Cendoroglo M (2006) Effects of uraemia and dialysis modality on polymorphonuclear cell apoptosis and function. Nephrol Dial Transplant 21:160–165
Lim WH, Kireta S, Leedham E, Russ GR, Coates PT (2007) Uremia impairs monocyte and monocyte-derived dendritic cell function in hemodialysis patients. Kidney Int 72:1138–1148
Vogtlander NP, Brown A, Valentijn RM, Rimmelzwaan GF, Osterhaus AD (2004) Impaired response rates, but satisfying protection rates to influenza vaccination in dialysis patients. Vaccine 22:2199–2201
Buti M, Viladomiu L, Jardi R, Olmos A, Rodriguez JA, Bartolome J, Esteban R, Guardia J (1992) Long-term immunogenicity and efficacy of hepatitis B vaccine in hemodialysis patients. Am J Nephrol 12:144–147
Krutzik SR, Hewison M, Liu PT, Robles JA, Stenger S, Adams JS, Modlin RL (2008) IL-15 links TLR2/1-induced macrophage differentiation to the vitamin D-dependent antimicrobial pathway. J Immunol 181:7115–7120
Baeke F, van Etten E, Gysemans C, Overbergh L, Mathieu C (2008) Vitamin D signaling in immune-mediated disorders: evolving insights and therapeutic opportunities. Mol Aspects Med 29:376–387
Overbergh L, Decallonne B, Valckx D, Verstuyf A, Depovere J, Laureys J, Rutgeerts O, Saint-Arnaud R, Bouillon R, Mathieu C (2000) Identification and immune regulation of 25-hydroxyvitamin D-1-alpha-hydroxylase in murine macrophages. Clin Exp Immunol 120:139–146
Vidal M, Ramana CV, Dusso AS (2002) Stat1-vitamin D receptor interactions antagonize 1,25-dihydroxyvitamin D transcriptional activity and enhance stat1-mediated transcription. Mol Cell Biol 22:2777–2787
Gombart AF, Borregaard N, Koeffler HP (2005) Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3. FASEB J 19:1067–1077
Chun RF, Adams JS, Hewison M (2008) Back to the future: a new look at ‘old’ vitamin D. J Endocrinol 198:261–269
Gombart AF, Bhan I, Borregaard N, Tamez H, Camargo CA Jr, Koeffler HP, Thadhani R (2009) Low Plasma Level of Cathelicidin Antimicrobial Peptide (hCAP18) Predicts Increased Infectious Disease Mortality in Patients Undergoing Hemodialysis. Clin Infect Dis 48:418–424
Andrukhova O, Zeitz U, Goetz R, Mohammadi M, Lanske B, Erben RG (2012) FGF23 acts directly on renal proximal tubules to induce phosphaturia through activation of the ERK1/2-SGK1 signaling pathway. Bone 51:621–628
Edfeldt K, Liu PT, Chun R, Fabri M, Schenk M, Wheelwright M, Keegan C, Krutzik SR, Adams JS, Hewison M, Modlin RL (2010) T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc Natl Acad Sci U S A 107:22593–22598
Bacchetta J, Sea J, Chun R, Lisse T, Wesseling-Perry K, Gales B, Adams J, Salusky IB, Hewison M (2012) FGF23 inhibits extra-renal synthesis of 1-25-dihydroxyvitamin D in human monocytes. J Bone Miner Res doi: 10.1002/jbmr.1740
Acknowledgments
This work was supported in part by educational grants (Académie Française/Jean Walter Zellidja, Réunion Pédiatrique de la Région Rhône Alpes, Société Française de Pédiatrie/Evian, Fondation pour la Recherche Médicale, Philippe Foundation, JB), by a grant from the Center for D-receptor Activation Research (CeDAR, MH), by USPHS grants DK 67563, DK 35423, DK 080984-01A1, RR 0333176–01 and funds from the Casey Lee Ball Foundation (IBS).
The authors would like to thank Katherine Wesseling-Perry, MD, Barbara Gales, RN, Joshua J Zaritsky, MD, PhD, and Renata C Pereira, PhD (Department of Pediatrics, David Geffen School of Medicine, University of California at Los Angeles, USA) for their helpful thoughts on FGF23 physiology, as well as Rene F Chun, PhD, Thomas S Lisse, PhD, and Jessica L Sea (Orthopaedic Research Hospital, University of California at Los Angeles, USA) for their technical help.
Disclosure of interests
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bacchetta, J., Salusky, I.B. & Hewison, M. Beyond mineral metabolism, is there an interplay between FGF23 and vitamin D in innate immunity?. Pediatr Nephrol 28, 577–582 (2013). https://doi.org/10.1007/s00467-012-2336-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-012-2336-0